10.07.2016 - 15.07.2016 in Girona, Spain
Pan-transglutaminase peptide microarray for high resolution epitope mapping
Katrin Bott-Fischer1, Volker Stadler2, Ralf Pasternack1 and Martin Hils1
1 Zedira GmbH, Rösslerstrasse 83, 64293 Darmstadt, Germany; 2 PEPperPRINT, Rischerstrasse 12, 69123 Heidelberg, Germany
Introduction
In the human body 8 transglutaminase (TG) iso-enzymes are described, fulfilling a variety of physiological functions. Poly- and monoclonal antibodies are important research tools for the elucidation of transglutaminase function in physiology and pathophysiology. Furthermore autoantibodies e.g. to TG2, TG3 or TG6 may be present in gluten-sensitive disorders and are used as diagnostic biomarkers.
PEPperCHIP® Transglutaminase Peptide Microarray is based on PEPperPRINT's unique peptide microarray platform, were peptides are synthesized on a coated glass slide upon printing of amino acid microparticles with a custom 24-color laser printer.
FXIIIA, TG1, TG2, TG3, TG4 and TG6 sequences were translated into 15 aa peptides printed in duplicate with a maximum peptide-peptide overlap of 14 aa for full epitope coverage. While the initial array presented here was limited to 6 transglutaminases, in the meantime chips including all transglutaminases (plus TG5, TG7 and non-enzymatic TG EPB4.2) are available.
Here we present a peptide microarray which allows high resolution epitope mapping of monoclonal (mabs) and polyclonal antibodies (pabs) for 6 transglutaminases in a single experiment. Further the method has the potential for patients‘ sera characterization.
Polyclonal abs and domain-specific mabs to TG2
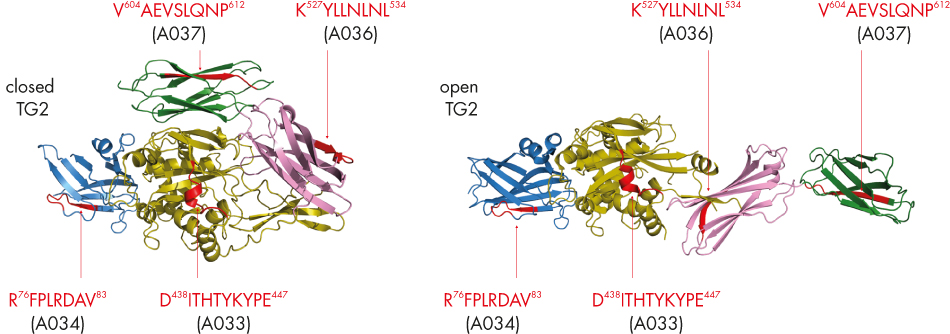
Epitope mapping of 4 monoclonal antibodies raised against full length human TG2 and characterized for their domain-specificity by western blotting using recombinantly produced TG2-domains has been performed using the PEPperCHIP® Transglutaminase Peptide Microarray. Incubation with the mabs at 1 -10 μg/mL followed by staining with control and secondary antibodies as well as fluorescence read out. Epitope mapping of polyclonal rabbit anti-human TG2 antibody A014 has been performed accordingly.
Fluorescence images of the microarray are shown in Fig. 1 for beta-sheet domain specific mab A034 and polyclonal ab A014. Fig. 2 summarizes the results for the four TG2-domain-specific mabs. Clear signals restricted to TG2-epitopes could be found for all mabs as well as for pab A014. Cross-reactivity to other transglutaminase isoenzymes has not been observed, confirming specificity data obtained by Western-blotting (data not shown). Thus the array proved its suitability for linear epitope-mapping of monoclonal and polyclonal antibodies.
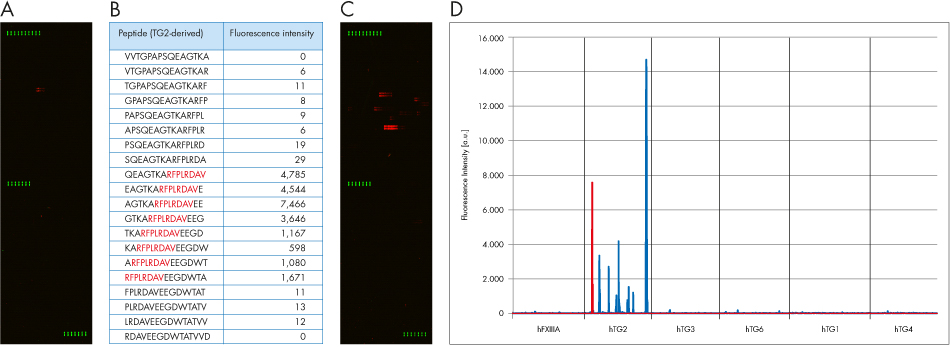
Figure 1: A) PEPperCHIP® Transglutaminase Peptide Microarray after incubation with mab A034 (1 μg/mL) and secondary anti-mouse IgG-antibody (DyLight680; red). Control: monoclonal anti-HAantibody-DyLight800; green). A weak but clear monoclonal response with a single epitope-like spot pattern formed by adjacent hTG2 peptides as well as a well-defined staining of HA control peptides (green) on top, in the middle and on bottom of the microarray is seen. B) Fluorescence intensity of the single spots around the A034 consensus motif RFPLRDAV (red). C) Microarray for pab A014 (10 μg/mL) revealing 10 clear epitope-like spot patterns with weak to strong intensity. Epitopes are indicated in fig 5. D) Fluorescence intensity plots for the whole microarray (red: mab A034, blue: pab A014).
Celiac disease patients‘ sera
In an initial attempt we analyzed 3 celiac disease patients’ sera with medium to high anti-TG2-IgA -titer by PEPperCHIP® Transglutaminase Peptide Microarray. The intensity plots reveal binding not only to TG2-epitopes, but also to epitopes of all other transglutaminases. In addition to conserved epitopes also serum-specific epitopes are given. Further each serum is characterized by its own intensity pattern. Fig. 3 shows two exemplary intensity plots.
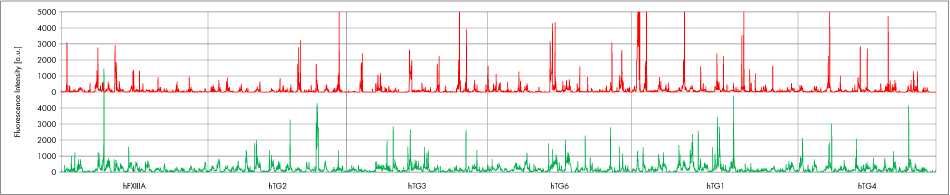
Figure 3: Fluorescence intensity plot for celiac disease patients’ sera (red: serum 283; green: serum 049).
Studies with non-celiac control sera are so far lacking, therefore an arbitrary serum specific cut-off was defined. Signal intensities above the cut-off were considered as an epitope. Based on aligned TG-sequences epitopes recognized by IgA autoantibodies of serum 049 were plotted in Fig. 4.
Common epitope regions can be deduced especially in the beta-sheet and core domain of the transglutaminases, interrupted by epitope free regions, e.g. around catalytic site cysteine. Increasing the arbitrarily chosen cut-off reduces the number and length of epitope-regions, but does not change the overall picture significantly. Thus celiac disease patients’ sera antibodies seem to be highly cross-reactive to linear epitopes of transglutaminase isoenzymes. It remains to be proven if this feature can be reproduced by ELISAs using full-length TG antigens.
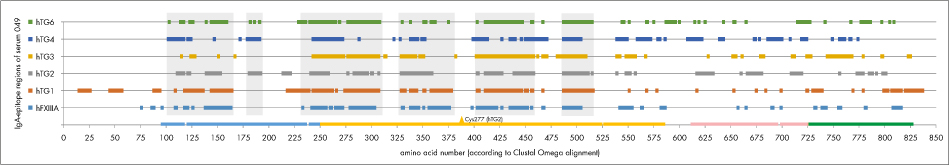
Figure 4: IgA-epitope regions for serum 049 deduced from the intensity plot using 200 a.u. as arbitrary cut-off plotted to aligned transglutaminase sequences (alignment by Clustal Omega). Common epitope regions are highlighted by grey rectangles. TG2-domains at the bottom line are coloured as in fig. 2.
Finally we plotted IgA and IgG-type anti-TG2-epitope regions defined according to fig. 4 for all sera (fig.5). Again conserved epitope regions can be deduced. Interestingly, epitopes for domain specific mabs as well as for pab A014 match to those conserved epitope regions. Antibodies did not bind to catalytic site Cys277 and its neighboring amino acids, while the other catalytic triad amino acids (His335 and Asp358) are part of epitope regions. For the 3 sera analyzed we found autoantibody binding epitopes all over the TG2-sequence. Contrarily, Iversen et al. (J. Immunol. 2013; 190:5981-5991) reported selective binding to the beta-sheet or catalytic domain only for 57 patient-mucosa derived monoclonal TG2-autoantibodies.
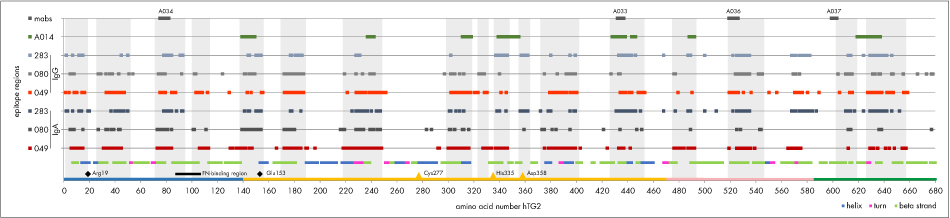
Figure 5: IgA-and IgG-epitope regions for 3 celiac disease patients‘ sera. In addition epitopes for domain specific mabs A033, A034, A036 and A037 as well as pab A014 are indicated. Common epitope regions are highlighted by rectangles. TG2-domains at the bottom line are coloured as in fig. 2.
The analyses of celiac disease patients’ sera using the microarray allow a novel patient specific insight into the autoantibody population. So far the data basis is not sufficient in order to draw conclusions or to discuss differences to published data. Furthermore one has to keep in mind, that non-linear, conformation and posttranslational modification specific epitopes cannot be detected by the microarray.
Summary and Conclusion
PEPperCHIP® Transglutaminase Peptide Microarray is a suitable tool for linear epitope mapping of monoclonal and polyclonal antibodies. The method bears the potential to be suitable for characterization of gluten sensitive patients' sera.
 Successful ISO9001:2015 recertification
Successful ISO9001:2015 recertification  Besuch des Bundesministers für Wirtschaft und Klimaschutz Dr. Robert Habeck bei der Zedira
Besuch des Bundesministers für Wirtschaft und Klimaschutz Dr. Robert Habeck bei der Zedira  Discover Our New Catalogue Edition and Dive into the World of Transglutaminases!
Discover Our New Catalogue Edition and Dive into the World of Transglutaminases!  Successful ISO9001:2015 recertification
Successful ISO9001:2015 recertification  Dr. Falk Pharma and Zedira announce successful completion of the phase 2a proof-of-concept study of ZED1227 for the treatment of Celiac Disease
Dr. Falk Pharma and Zedira announce successful completion of the phase 2a proof-of-concept study of ZED1227 for the treatment of Celiac Disease  Dr. Falk Pharma und Zedira verkünden den erfolgreichen Abschluss der Phase 2a-Studie mit ZED1227 zur Behandlung von Zöliakie
Dr. Falk Pharma und Zedira verkünden den erfolgreichen Abschluss der Phase 2a-Studie mit ZED1227 zur Behandlung von Zöliakie  Reversibly acting transglutaminase 2 inhibitors: drug candidates for the treatment of fibrosis
Reversibly acting transglutaminase 2 inhibitors: drug candidates for the treatment of fibrosis  Transcriptomic analysis of the efficacy of TG2-inhibitor trials and human intestinal organoids modelling Celiac disease pathogenesis
Transcriptomic analysis of the efficacy of TG2-inhibitor trials and human intestinal organoids modelling Celiac disease pathogenesis  Transglutaminase antibodies and neurological manifestations of gluten sensitivity
Transglutaminase antibodies and neurological manifestations of gluten sensitivity  Design of Oral FXIIIa Blockers as Safer Anticoagulants Mission Impossible?
Design of Oral FXIIIa Blockers as Safer Anticoagulants Mission Impossible?  Microbial transglutaminase (MTG) enables efficient and site-specific conjugation to native antibodies without the need of antibody engineering
Microbial transglutaminase (MTG) enables efficient and site-specific conjugation to native antibodies without the need of antibody engineering  Tridegin as FXIIIa inhibitor
Tridegin as FXIIIa inhibitor  Microbial transglutaminase: from discovery to market
Microbial transglutaminase: from discovery to market  Tissue transglutaminase inhibitors
Tissue transglutaminase inhibitors  Tissue transglutaminase in Alzheimers Disease
Tissue transglutaminase in Alzheimers Disease  Factor XIIIa: novel target for anticoagulation?
Factor XIIIa: novel target for anticoagulation?  Microbial transglutaminase for site-specific protein conjugation
Microbial transglutaminase for site-specific protein conjugation