In the human body, eight transglutaminase isoenzymes (TG1-TG7 and coagulation factor XIII) fulfill a broad variety of physiological functions. However, transglutaminases are also involved in the pathophysiology of various disorders.
In celiac disease, Zedira’s drug candidate ZED12271, an irreversible direct-acting and specific inhibitor of human tissue transglutaminase (TG2), showed proof of concept in a phase 2a study. The groundbreaking results have been published in the New England Journal of Medicine. An overview is given in our press release.
This proof of concept is of utmost importance not only for celiac disease but beyond. For the first time, a transglutaminase was validated as a druggable target. Moreover, our transglutaminase inhibitor ZED1227 was proven to be safe and well tolerated.
This remarkable success has a favorable effect on several other transglutaminase-related disorders. In particular, fibrotic disorders affecting the lung, liver, or kidney may be addressed with our compounds. Our newest class of reversible direct-acting and specific inhibitors of tissue transglutaminase (TG2) are orally bioavailable proprietary compounds. An overview is given in our press release.
Transglutaminase blocker drug candidates are also available to target coagulation factor XIII (F13, plasma transglutaminase). This novel and unique approach dissociates the antithrombotic effect from the increased bleeding tendency in anticoagulation. Results obtained in an in vivo study of the direct-acting factor XIIIa inhibitor ZED3197 have been published in the Journal of Thrombosis and Haemostasis. For detailed information, please refer to the respective open-access scientific publication.
Celiac Disease
Zedira’s drug candidate ZED1227 targeting dysregulated TG2 in celiac disease showed proof of concept in a phase 2a clinical trial. The compound has been licensed to Dr. Falk Pharma and Takeda Pharmaceuticals.
read more
Gluten Sensitive Neurological Disorders
Autoantibodies specific for neuronal transglutaminase (TG6) can be measured with our proprietary ELISA kits.
New: Analytical service for measurement of TG6-autoantibodies in human serum samples.
read more
Thrombosis/Anticoagulation
Proof-of-principle for safe anticoagulation, meaning anticoagulation without increased bleeding risk, using blood coagulation factor XIIIa-blockers has recently been shown.
read more
1 Büchold C, Hils M, Gerlach U, Weber J, Pelzer C, Heil A, Aeschlimann D, Pasternack R. Features of ZED1227: The First-In-Class Tissue Transglutaminase Inhibitor Undergoing Clinical Evaluation for the Treatment of Celiac Disease. Cells. 2022; 11(10):1667. https://doi.org/10.3390/cells11101667
 Besuch des Bundesministers für Wirtschaft und Klimaschutz Dr. Robert Habeck bei der Zedira
Besuch des Bundesministers für Wirtschaft und Klimaschutz Dr. Robert Habeck bei der Zedira  Discover Our New Catalogue Edition and Dive into the World of Transglutaminases!
Discover Our New Catalogue Edition and Dive into the World of Transglutaminases!  Successful ISO9001:2015 recertification
Successful ISO9001:2015 recertification  Dr. Falk Pharma and Zedira announce successful completion of the phase 2a proof-of-concept study of ZED1227 for the treatment of Celiac Disease
Dr. Falk Pharma and Zedira announce successful completion of the phase 2a proof-of-concept study of ZED1227 for the treatment of Celiac Disease  Dr. Falk Pharma und Zedira verkünden den erfolgreichen Abschluss der Phase 2a-Studie mit ZED1227 zur Behandlung von Zöliakie
Dr. Falk Pharma und Zedira verkünden den erfolgreichen Abschluss der Phase 2a-Studie mit ZED1227 zur Behandlung von Zöliakie  Reversibly acting transglutaminase 2 inhibitors: drug candidates for the treatment of fibrosis
Reversibly acting transglutaminase 2 inhibitors: drug candidates for the treatment of fibrosis  Transglutaminase antibodies and neurological manifestations of gluten sensitivity
Transglutaminase antibodies and neurological manifestations of gluten sensitivity  Design of Oral FXIIIa Blockers as Safer Anticoagulants Mission Impossible?
Design of Oral FXIIIa Blockers as Safer Anticoagulants Mission Impossible?  Microbial transglutaminase (MTG) enables efficient and site-specific conjugation to native antibodies without the need of antibody engineering
Microbial transglutaminase (MTG) enables efficient and site-specific conjugation to native antibodies without the need of antibody engineering 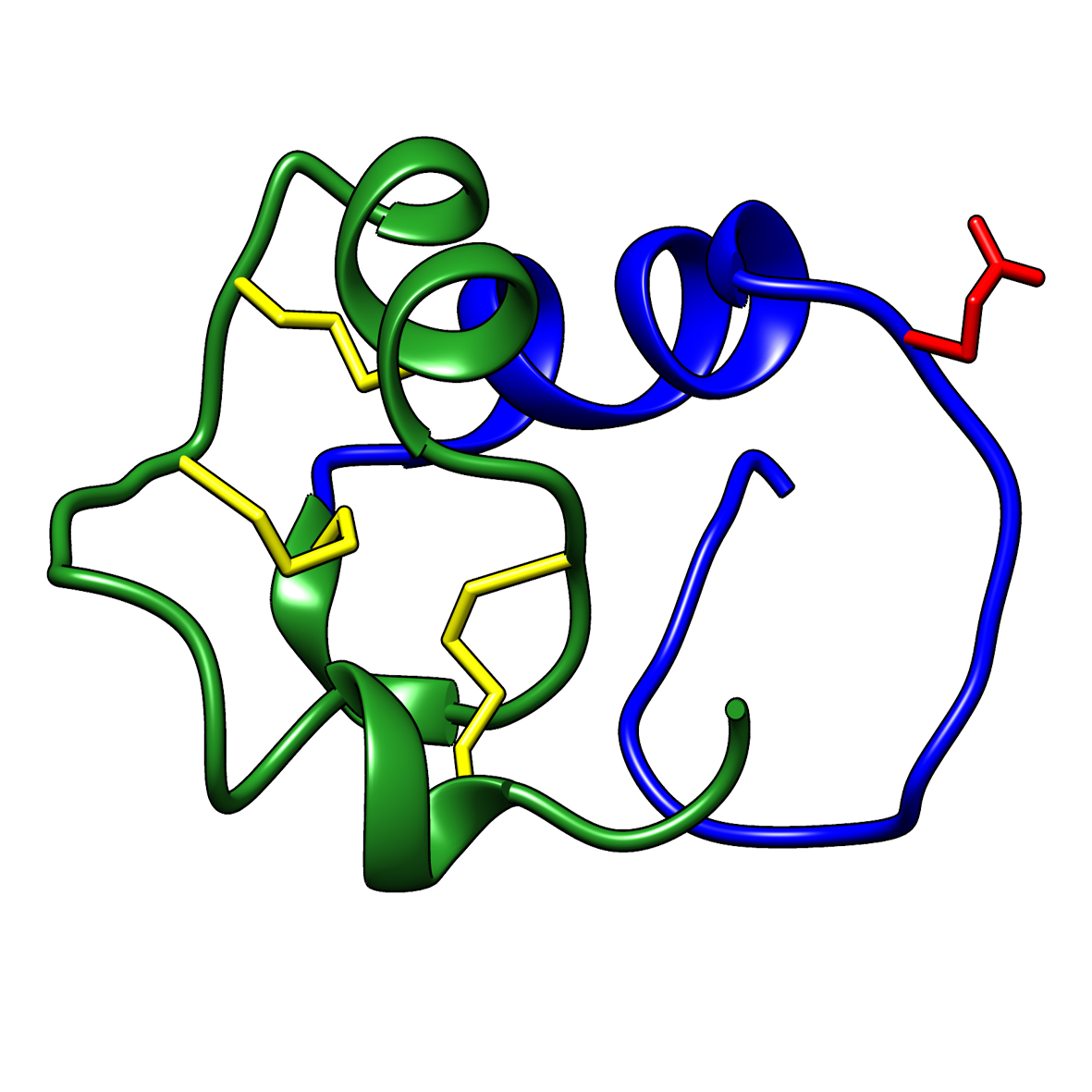 Tridegin as FXIIIa inhibitor
Tridegin as FXIIIa inhibitor 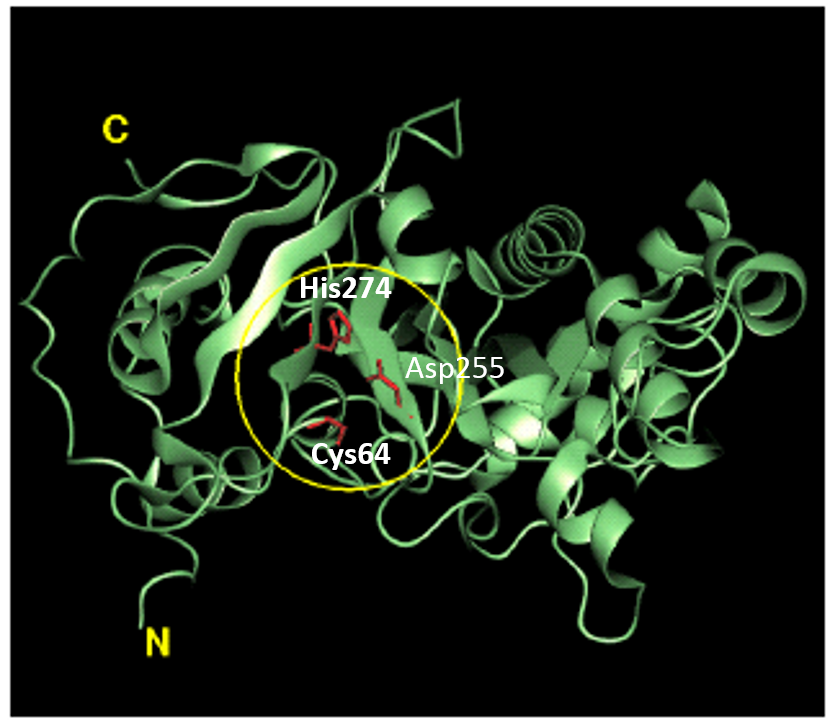 Microbial transglutaminase: from discovery to market
Microbial transglutaminase: from discovery to market  Tissue transglutaminase inhibitors
Tissue transglutaminase inhibitors  Tissue transglutaminase in Alzheimers Disease
Tissue transglutaminase in Alzheimers Disease  Factor XIIIa: novel target for anticoagulation?
Factor XIIIa: novel target for anticoagulation? 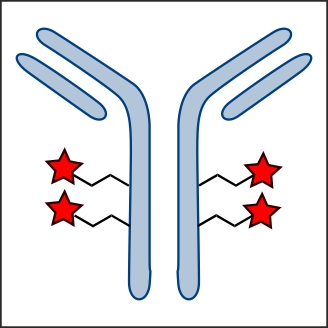 Microbial transglutaminase for site-specific protein conjugation
Microbial transglutaminase for site-specific protein conjugation